- The posting you are trying to access is no longer available for comment.
Bulletin Board
Below are two lists:
- The first list displays policies and procedures that have recently been approved and/or came into effect in the last two months.
- The second list displays draft documents that are open for consultation/comment. The
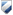 indicates that a document is only available for comment to select individuals (e.g. select staff, staffing groups, service types, advisory group members). A password will be needed to access these documents. Passwords are issued by the policy author. Draft documents will generally be available for comment to staff for at least 10 working days. Draft documents should not be construed as approved documents nor relied upon in any way.
indicates that a document is only available for comment to select individuals (e.g. select staff, staffing groups, service types, advisory group members). A password will be needed to access these documents. Passwords are issued by the policy author. Draft documents will generally be available for comment to staff for at least 10 working days. Draft documents should not be construed as approved documents nor relied upon in any way.
Feedback and comment on existing documents may be provided at any time directly to the Policy Author. Policy Author contact details are recorded on the 'Status and Details' page. Simply open the document, and then click the link to the 'Status and Details' page (located at top of page on the light grey menu bar).
Recent Approvals
Bulletin Board Postings
Document Title
Date Due for Comment
Document Author
There are no postings to display.